In this article
Picture this: you’ve been feeling off for months. Tired in a way that sleep doesn’t fix. Gaining weight despite eating well. Cold when everyone around you is perfectly comfortable. Brain fog that makes it hard to remember basics. You finally get answers — Hashimoto’s thyroiditis — and your first question is why me?
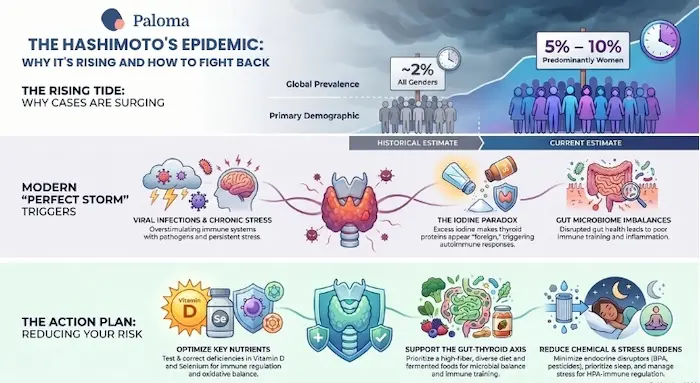
You’re far from alone in asking. Hashimoto’s thyroiditis, an autoimmune condition in which your immune system attacks your thyroid gland, has transformed from a relatively obscure diagnosis into what many endocrinologists now call a modern epidemic. Just a decade ago, estimates placed the global prevalence at around 2%. Today, Hashimoto’s affects somewhere between 5 and 10% of the population — and some regional studies put that number even higher. That’s tens of millions of people, the vast majority of them women.
The numbers are rising too fast to be explained away by genetics, better detection, or coincidence. Something in the fabric of modern life is triggering immune systems to misfire — and science is getting closer to understanding exactly what that is.
Before diving into why Hashimoto’s disease is on the rise, it helps to understand what’s happening inside the body. Hashimoto’s is an autoimmune disorder, meaning the immune system — which is supposed to protect you — mistakenly identifies your thyroid gland as a threat and begins attacking it. Over time, this chronic immune assault damages the thyroid’s ability to produce the thyroid hormones your body depends on for energy metabolism, temperature regulation, mood, cognition, and more.
The condition is defined by the presence of specific autoantibodies: thyroid peroxidase antibodies (TPOAb) and thyroglobulin antibodies (TgAb). TPOAb is positive in roughly 95% of people with Hashimoto’s, making it the cornerstone of diagnosis alongside thyroid ultrasound. But here’s the catch — a person can test positive for these antibodies for years, even decades, before their thyroid function visibly declines. That silent, subclinical period is one reason why Hashimoto’s so often goes undetected until symptoms become hard to ignore.
It’s also why the rising prevalence numbers may still be an undercount.
Researchers are increasingly clear that genetic factors alone don’t explain the epidemic. While certain gene variants — particularly in the immune system genes — do raise susceptibility, genes don’t change fast enough to account for a prevalence that has more than doubled in recent decades. The real drivers are a variety of triggers – environmental, behavioral, and systemic – and many of them are distinctly modern.
The iodine paradox
Iodine is essential for life. Your thyroid literally cannot make thyroid hormones without it. In the mid-20th century, iodized salt programs were introduced across much of the world to combat widespread iodine deficiency. This genuine public health success dramatically reduced conditions like goiter and congenital hypothyroidism (previously known as cretinism).
But the story has an ironic twist. Research now shows that too much iodine can be just as problematic for thyroid health as too little. When thyroglobulin — the protein that helps store and release thyroid hormones — becomes excessively iodinated, it becomes foreign to the immune system, triggering an autoimmune response. Excess iodine also increases oxidative stress in the thyroid and promotes the death of thyroid follicular cells, essentially handing the immune system reasons to attack.
This helps explain why Hashimoto’s rates are highest in iodine-sufficient and iodine-excess regions. As fortified foods, iodine-rich supplements, and processed food diets have become a global norm, the thyroid-autoimmune connection has followed closely behind.
The takeaway isn’t to eliminate iodine — far from it. But it does underscore that more isn’t always better, and that hidden iodine in supplements and processed foods deserves attention.
Viral triggers: From EBV to COVID-19
.webp)
Your immune system is designed to fight infections. But in people with genetic susceptibility, certain viruses appear to leave a lasting, damaging footprint on thyroid immunity. The mechanism is called molecular mimicry — in essence, a viral protein looks similar enough to a thyroid protein that the antibodies your immune system makes to fight the infection also start targeting your thyroid. Once that response is activated, it can become self-perpetuating.
Epstein-Barr virus (EBV), the virus responsible for mononucleosis, has long been the most studied culprit. EBV infects B cells — the very immune cells that produce antibodies — and evidence suggests it can reactivate immune targeting of thyroid tissue in susceptible individuals. Human herpesvirus 6, parvovirus B19, and hepatitis C have all been implicated as well.
Then came SARS-CoV-2. Research published since 2020 has confirmed a meaningful link between COVID-19 infection and new-onset thyroid dysfunction, including Hashimoto’s. COVID-19 is known to trigger significant and prolonged immune dysregulation, and the spike protein has been studied for potential cross-reactivity with thyroid antigens. The full downstream impact of the pandemic on autoimmune thyroid disease rates is still unfolding, but thyroid specialists are watching closely.
For patients, this underscores a critical point: a Hashimoto’s diagnosis that follows a significant illness is not a coincidence. It is a pattern the research increasingly supports.
The gut-thyroid connection
One of the most fascinating and rapidly evolving areas of Hashimoto’s research is the relationship between gut health and thyroid autoimmunity. The gut microbiome — the vast ecosystem of bacteria, viruses, and fungi that lives in your digestive tract — plays a central role in regulating immune function. When that ecosystem is disrupted, the immune system can lose its ability to distinguish between self and non-self.
Research has found that people with Hashimoto’s and an underactive thyroid tend to have measurable differences in their gut microbiome compared to healthy controls, including reduced populations of beneficial bacteria like Bifidobacterium and an altered balance of immune-signaling microbes. The gut and the thyroid also communicate through what scientists are calling the “gut-thyroid axis” — a bidirectional relationship in which gut microbiota affects thyroid hormone metabolism, iodine uptake, and TSH release. Thyroid status, in turn, influences microbial composition.
The culprits behind microbiome disruption are largely modern: overuse of antibiotics, highly processed diets low in fiber, chronic stress, reduced contact with diverse natural environments, and early childhood exposures all reshape the gut ecosystem in ways that appear to raise autoimmune risk. There is even a concept called the “hygiene hypothesis” — the idea that as developed-world environments have become cleaner and less microbially diverse, immune systems have lost the training they need to behave properly, turning inward instead.
Nutritional deficiencies: Vitamin D and selenium
Two nutrients in particular keep appearing in Hashimoto’s research as protective factors that are increasingly absent from modern diets and lifestyles.
Vitamin D is less a vitamin and more a hormone, playing a direct role in immune system regulation. Vitamin D receptors are found on immune cells throughout the body, and adequate vitamin D helps keep inflammatory and autoimmune responses in check. Deficiency — which is now genuinely widespread due to reduced sun exposure, indoor lifestyles, and widespread use of sunscreen — has been consistently associated with higher rates of autoimmune thyroid disease. Some studies have found that people with Hashimoto’s are significantly more likely to be vitamin D-deficient than the general population.
Selenium is a trace mineral that is essential for thyroid function. The thyroid has one of the highest concentrations of selenium of any organ in the body, and it relies on selenium-dependent enzymes to produce thyroid hormones, neutralize oxidative stress, and protect thyroid cells from damage. Selenium deficiency has been linked to elevated thyroid antibodies and worsened disease activity in Hashimoto’s patients. As selenium levels in soil vary widely — and food processing tends to reduce selenium content — many people are quietly deficient without knowing it.
Environmental chemicals and endocrine disruptors

Modern life comes with an invisible chemical burden that the thyroid is particularly vulnerable to. The thyroid gland is extremely sensitive to environmental compounds and chemicals that can disrupt hormone production and immune function, including:
- PCBs and polybrominated diphenyl ethers (PBDEs), found in flame retardants, electronics, and industrial contamination, bind to thyroid transport proteins and mimic or block thyroid hormone action
- Pesticides and herbicides, including glyphosate, have been studied for potential effects on both gut microbiota and thyroid function
- Heavy metals such as cadmium and mercury, which can accumulate in thyroid tissue and alter its function
- Bisphenol A (BPA) and similar plasticizers found in food packaging have demonstrated thyroid-disrupting properties in laboratory studies
These chemicals are virtually everywhere in developed-world environments. Their effects on any individual are difficult to quantify. Still, population-level exposure has increased dramatically over the past several decades — roughly in parallel with the rise in autoimmune diseases, including thyroid disease. Researchers describe them as “co-triggers” that interact with genetics and environmental factors to push the immune system over the edge.
Chronic stress and the HPA-immune axis
Stress doesn’t just feel bad — it actively reshapes your immune function. The body’s stress response, mediated through the hypothalamic-pituitary-adrenal (HPA) axis and cortisol release, is designed for short-term crises. When stress becomes chronic, cortisol’s ability to regulate immune activity degrades. The result can be a shift toward the kind of dysregulated, pro-inflammatory immune activity that underlies autoimmune conditions.
Thyroid clinicians frequently observe that Hashimoto’s disease flares or new diagnoses follow periods of sustained emotional or physical stress — grief, major illness, job loss, significant life upheaval. This is not anecdotal. Research supports the view that stress is a meaningful environmental trigger in genetically susceptible individuals. The modern epidemic of chronic psychological stress, compounded by inadequate sleep and sedentary lifestyles, adds another layer to an already complex picture.
The diagnosis effect: More testing, more cases found
It would be incomplete to discuss the rising prevalence of Hashimoto’s without acknowledging that some of the increase reflects improved detection. Thyroid antibody testing has become more routine, ultrasound technology has improved, and there is broader awareness of the condition among both patients and clinicians. Cases that would have quietly gone undiagnosed a generation ago are now being identified.
However, researchers are clear that improved detection alone does not explain the magnitude of the increase. The rise in both incidence and prevalence is real, and it is happening across populations with very different healthcare access and testing habits. The environmental and lifestyle forces described above are genuinely increasing the number of people whose immune systems are misfiring against their thyroid, not just increasing the number of people whose existing disease is being found.
Let’s be honest here: Hashimoto’s disease cannot always be prevented. If you have strong genetic risk factors, no amount of dietary optimization will guarantee that you never develop the condition. But the science does support several meaningful steps that may reduce your risk, delay onset, or slow progression — particularly for those of you who know you have elevated antibodies but have not yet developed hypothyroidism.
Think of these not as guarantees, but as tools worth having.
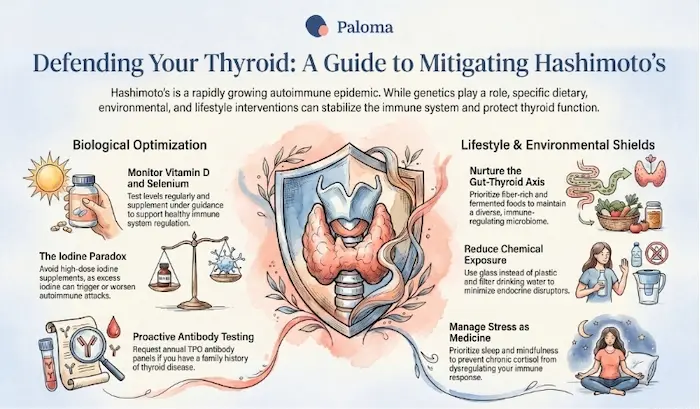
Check your vitamin D and selenium levels
This is the single most actionable step for most people. Both vitamin D and selenium have well-documented immune-regulatory roles in thyroid health, both deficiencies are common and correctable, and supplementation — when guided by actual blood test results — is safe and accessible.
Ask your doctor to test your 25-OH vitamin D level and consider testing selenium as well. If you’re deficient, appropriate supplementation under medical guidance has been shown to reduce thyroid antibody levels and support overall immune regulation. Do not supplement with selenium at high doses without guidance, as excess selenium is harmful.
Be mindful of iodine intake
You need iodine — but you need the right amount. Avoid high-dose iodine supplements unless specifically directed by a physician. Be aware that kelp, seaweed, and some “thyroid support” supplements can contain iodine levels well above your daily needs. Iodized salt used in ordinary cooking is generally fine. The issue is with concentrated supplemental forms.
Protect and nurture your gut
A fiber-rich, diverse diet is the foundation of microbiome health. Prioritize vegetables, legumes, whole grains, and fermented foods. Minimize ultra-processed food, which is low in fiber and high in additives that can disrupt your gut bacteria. Use antibiotics only when medically necessary, and consider probiotic support after any antibiotic course. Emerging research on the gut-thyroid axis makes gut health one of the most promising modifiable targets for autoimmune thyroid risk.
Reduce chemical exposures where you can
Perfection is impossible here, but meaningful reductions are achievable. Choose glass or stainless steel over plastic for food and drink preparation and storage when practical. Wash produce thoroughly. Choose organic options for the highest-pesticide produce categories when your budget allows. Filter your drinking water. These steps won’t eliminate your chemical burden, but they can meaningfully reduce it over time.
Manage stress seriously

Keeping stress under control is easier said than done, but it deserves more than a footnote. Chronic stress is not just uncomfortable — it is immunologically active. Prioritize sleep with the same seriousness you’d give a prescription medication. Build genuine stress recovery practices into your routine in whatever form works best for you: movement, mindfulness, therapy, time in nature, or simply protecting unscheduled time. For people with known thyroid antibodies or a family history of Hashimoto’s, stress management is a necessary medical intervention, not a luxury.
Get appropriate monitoring if you’re at higher risk
If you have a first-degree relative with Hashimoto’s, have tested positive for thyroid antibodies, or have another autoimmune condition, proactive monitoring makes sense. This means an annual thyroid test panel, including antibody testing, discussion with your doctor about ultrasound monitoring, and attention to symptoms. Early identification gives you the best chance to intervene before significant thyroid damage occurs.
Note: The Paloma Complete Thyroid Test kit makes it easy to test thyroid-stimulating hormone (TSH), Free T4, Free T3, and Thyroid Peroxidase (TPO) antibodies easily and painlessly from home.
Hashimoto’s thyroiditis is rising because the modern world—for all its comforts—is placing new and compounding burdens on the immune systems of people who are genetically primed to be vulnerable. Too much iodine, too little vitamin D, a disrupted gut microbiome, constant exposure to endocrine-disrupting chemicals, chronic stress, and a growing list of immune-triggering infections have converged into a perfect storm for autoimmune thyroid disease.
None of this is cause for despair. It is a call for deeper understanding—and for taking the steps within our control seriously. The more we understand why Hashimoto’s is increasing, the better equipped patients and their healthcare teams are to push back against that tide.
That’s where having the right support matters. As a Paloma Health patient, you have access to thyroid-focused care that looks beyond a single lab value to the bigger picture—autoimmunity, symptoms, nutrition, and lifestyle. With convenient at-home testing, expert-guided treatment, and ongoing monitoring, Paloma helps you take a more proactive, personalized approach to managing Hashimoto’s.
If you’ve been diagnosed, you are part of a vast and growing community navigating this condition—and you don’t have to do it alone. And if you haven’t been diagnosed but recognize yourself in this picture, the most powerful thing you can do is get informed, get tested, and take action with a care team that understands the full complexity of thyroid health.

What is Hashimoto’s thyroiditis in simple terms?
It’s an autoimmune condition where your immune system mistakenly attacks your thyroid, eventually reducing its ability to produce essential hormones.
Why are so many more people being diagnosed with Hashimoto’s today?
The increase is linked to modern factors such as diet, environmental toxins, stress, infections, and gut health issues—not just to better testing.
Can you have Hashimoto’s without knowing it?
Yes, many people have thyroid antibodies for years without symptoms, which is why diagnosis is often delayed.
Does iodine help or hurt Hashimoto’s?
Your body needs iodine, but too much—especially from supplements—can actually trigger or worsen autoimmune thyroid activity.
How are viruses connected to Hashimoto’s?
Certain viruses may confuse the immune system through “molecular mimicry,” causing it to mistakenly attack thyroid tissue.
What role does gut health play in thyroid disease?
An unhealthy gut microbiome can disrupt immune regulation, increasing the risk of autoimmune conditions like Hashimoto’s.
Are there specific nutrient deficiencies linked to Hashimoto’s?
Yes—low vitamin D and selenium levels are commonly associated with higher thyroid antibody levels and worse disease activity.
Can stress really affect your thyroid?
Absolutely—chronic stress can dysregulate the immune system and is often linked to the onset or worsening of Hashimoto’s.
Is Hashimoto’s preventable?
Not always, especially if you have a genetic risk, but lifestyle changes can reduce your risk or slow progression.
What’s the most important first step if you’re concerned about Hashimoto’s?
Get tested—especially thyroid antibodies—and work with a knowledgeable provider to monitor and manage your thyroid health early.

.webp)



.webp)

%20(1).webp)






