In this article
It is one of the most common frustrations in hypothyroid patient communities, repeated so often across social media Reddit, TikTok, and Facebook groups that it has become something of a battle cry: “My doctor says my thyroid labs are fine, but I still feel terrible.”
The fatigue is relentless. The weight won’t move despite every effort. The brain fog makes simple tasks feel like advanced calculus. Your mood mood is low and you feel cold constantly. And the doctor keeps pointing at the TSH number on the lab report — normal, “in range” — and offers no further explanation or solutions.
If you are one of these hypothyroid patients, there’s an explanation. It’s not a mystery.
You have a T4-T3 conversion problem!
Levothyroxine, the synthetic T4 medication that has been the standard of care for hypothyroidism for decades, is not an active thyroid hormone. It’s actually a prohormone — a raw material. For levothyroxine to actually do something in your cells, your body must convert it into T3 (triiodothyronine), the biologically active thyroid hormone. This conversion is performed by a family of enzymes called deiodinases.
Here’s the challenge: In a significant portion of the population, this process is genetically impaired in ways that standard thyroid blood tests cannot detect.
Layer on top of that a second, increasingly documented problem: your gut microbiome plays a direct and measurable role in thyroid hormone metabolism. Most hypothyroidism in the U.S. is a result of Hashimoto’ thyroiditis, an autoimmune disease. And when the gut is imbalanced or your intestinal barrier is compromised — conditions that are remarkably common in people with autoimmune thyroid disease — the conversion and reabsorption of thyroid hormones can be further undermined.
This is the T3/T4 conversion problem. And it is why, for a meaningful subset of thyroid patients, taking standard levothyroxine treatment and achieving a normal thyroid-stimulating hormone (TSH) level is not the same thing as getting well!
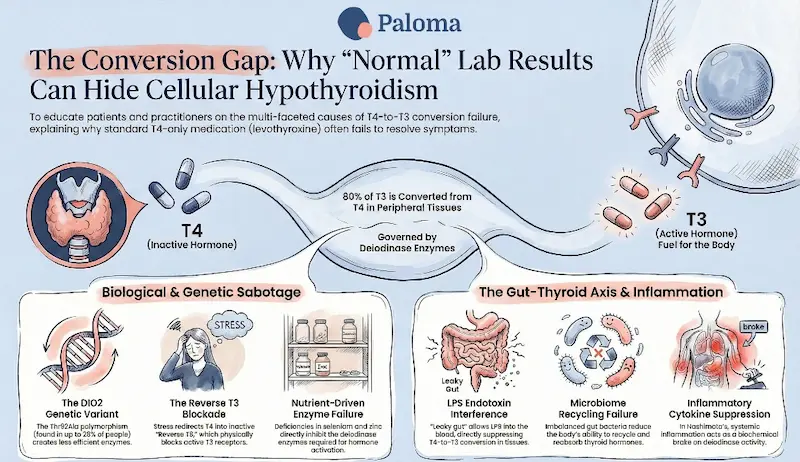
To understand the conversion problem, it helps to understand the basic biology. The thyroid gland primarily secretes T4 — thyroxine — a relatively inert storage form of thyroid hormone.
Less than 20% of the circulating T3 that your cells actually use comes directly from the thyroid gland itself. The rest — more than 80% — is produced outside the thyroid gland via conversion from T4. This conversion happens in the liver, kidneys, skeletal muscle, brain, and other tissues, and is governed primarily by type 1 and type 2 deiodinase enzymes (DIO1 and DIO2).
When you take levothyroxine, you are supplementing the T4 level. Your TSH will fall, your free T4 will rise, and on paper, everything looks fine. But whether that T4 is being efficiently converted into T3 in the tissues that need it — the brain, the heart, the muscles, the gut — is an entirely separate question. It is a question that a TSH test does not answer. And for patients with genetic variants in their deiodinase enzymes, the answer is often: not well enough!
Your TSH reflects whether your pituitary is satisfied. It says nothing about whether your brain, heart, and muscles are getting the T3 they need to function.
The most extensively studied genetic variant, or polymorphism, is in the DIO2 gene — the gene that encodes type 2 deiodinase, the enzyme responsible for T4-to-T3 conversion in key tissues including the brain, brown adipose tissue, skeletal muscle, and placenta.
This Thr92Ala polymorphism is found in 12 to 36% of the general population — meaning between roughly 1 in 8 and more than 1 in 3 people carry at least one copy. It is not rare. It is one of the most common functional genetic variants in human thyroid metabolism.
Research published in the Journal of Clinical Endocrinology & Metabolism provided direct clinical evidence of what this variant does: patients carrying the Thr92Ala polymorphism had significantly lower free T3 levels after thyroidectomy compared to non-carriers, despite comparable TSH levels. The variant reduced T4-to-T3 conversion, and levothyroxine treatment alone was not adequate to compensate. The researchers concluded that people with the Thr92Ala genetic variation are at increased risk of having reduced T3 levelss that cannot be corrected by standard levothyroxine replacement.
A landmark study, published in 2019 in the Journal of Clinical Investigation, found that another genetic variant – Ala92-D2 – creates a “mutant” enzyme that generates significantly less T3. When animals carrying this polymorphism were tested, they were found to be less physically active, they slept more, took longer to form memories, and had profiles consistent with cellular hypothyroidism — even when their circulating thyroid hormone levels appeared normal.
This is the essence of the T4-T3 conversion problem. Tissues are starved of active thyroid hormone while lab reports say otherwise. Patients are told they are fine. They are not.
The DIO2 variant is the most studied culprit, but it is not the only one. Thyroid hormone conversion is a multi-step process that can be disrupted at several other points as well.
Type 1 deiodinase (D1), primarily expressed in the liver and kidneys, produces a large proportion of circulating T3. D1 activity is sensitive to a range of stressors: chronic illness, caloric restriction, high cortisol (a common feature of chronic stress), selenium deficiency, and systemic inflammation can all suppress D1 activity and reduce T4-to-T3 conversion rates. This means that even a patient without the DIO2 polymorphism can end up with inadequate T3 if they are chronically stressed, poorly nourished, or carrying high inflammatory load — all of which are disproportionately common in people with autoimmune thyroid disease.
There is also the reverse T3 problem. Under conditions of physiological stress, the body can redirect T4 conversion away from active T3 and toward reverse T3 — an inactive form of T3 that competes with T3 for cellular receptors. A high reverse T3 effectively blocks the action of whatever T3 is present. Standard thyroid function panels do not measure reverse T3 and most conventional physicians do not order it. But for patients with persistent symptoms despite normal TSH and T4, elevated reverse T3 can be a significant and overlooked part of the picture.
If the DIO2 story is the chapter thyroid patients are just beginning to understand, the gut-thyroid axis is a chapter most have never even considered heard. Research is now making it unmistakably clear: the gut microbiome is not a passive bystander in thyroid hormone metabolism. It is an active participant.
A 2022 review in Frontiers in Endocrinology explained how the gut plays at least three distinct roles in thyroid hormone handling:
- Deiodinase activity is present in the intestinal wall, and gut bacteria directly affect it. Research in animal models has found that intestinal deiodinase activity — the gut’s own ability to convert T4 to T3 — is suppressed by the resident bacteria. When beneficial bacterial populations decline, this regulation is disrupted and negatively affects conversion.
- The gut microbiome manages the recycling of thyroid hormones. In the gut, bacteria break down thyroid hormone metabolites and free them up to be reabsorbed. When there’s an imbalance in gut bacteria, less thyroid hormone is recovered, and more is lost in the stool.
- Lipopolysaccharide (LPS) — an endotoxin produced by bacteria that leaks into circulation when your intestinal barrier integrity is compromised (also known as “leaky gut”) — directly interferes with deiodinase activity. LPS inhibits DIO1, the enzyme responsible for producing the bulk of circulating T3. At the same time, it activates DIO2, which can suppress TSH output from the pituitary even while peripheral tissues remain T3-deficient. The result is that while lab values look better, the patient feels worse.
For patients with Hashimoto’s thyroiditis specifically, the conversion problem is compounded by their underlying autoimmune condition in ways that deserve special attention.
Research has shown that the DIO2 Thr92Ala polymorphism is one of the primary reasons why a substantial proportion of Hashimoto’s patients continue to experience symptoms, despite achieving normal thyroid levels. The research notes that while this variant is found in 12 to 36% of the general population, it’s a genetic vulnerability that is completely invisible to standard TSH-based monitoring.
The autoimmune process of Hashimoto’s also drives systemic inflammation via pro-inflammatory cytokines. These inflammatory signals directly suppress deiodinase activity and can impair T3 conversion, even in patients who do not carry the DIO2 polymorphism. The autoimmune disease itself is, in effect, biochemically sabotaging the conversion process.
And Hashimoto’s patients have significantly higher rates of gut dysbiosis and intestinal permeability. Research has consistently found reduced populations of beneficial bacteria in autoimmune thyroid disease, while populations of bacteria associated with intestinal inflammation are elevated. According to researchers, this dysbiosis creates a hostile gut environment that undermines the job of thyroid hormone medication.
The result is a triple threat:
- a potential genetic impairment of conversion (DIO2)
- an inflammatory suppression of deiodinase activity
- imbalanced gut bacteria that reduce the effective dose of whatever thyroid hormone does get converted.
For many thyroid patients, the problem isn’t the medication. It’s a thyroid conversion system that’s been compromised at multiple levels — genetically, immunologically, and microbially.
1. Test beyond TSH
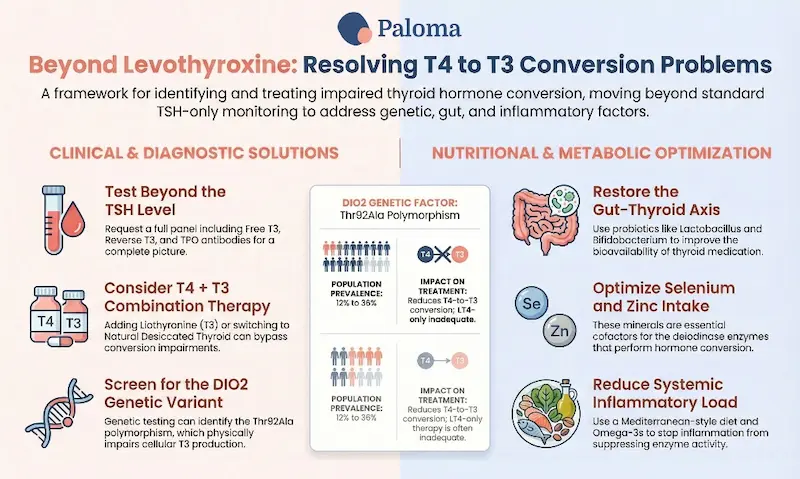
If you are taking levothyroxine and not feeling well, your first step is getting a comprehensive picture of your thyroid hormone status — not just TSH. A complete thyroid full panel should include free T3, free T4, reverse T3, and thyroid peroxidase (TPO) antibodies. (Note: The Paloma Complete Thyroid Test kit includes TSH, free T3, free T4, and TPO antibodies, and a reverse T3 can be included as well.) A low free T3 or high reverse T3 in the context of a normal TSH is a clear signal that the conversion pathway is not functioning optimally. Ask your provider to run these tests. If they decline, thyroid-focused providers like the team of experts at Paloma will work with you to optimize your hypothyroidism treatment.
You can also pursue genetic testing. DNA health panels from 23 and Me and Ancestry can identify whether you have thyroid-related polymorphisms.
2. Consider T4+T3 combination therapy
For patients with documented low free T3 and persistent symptoms despite adequate levothyroxine, combination therapy adding liothyronine (synthetic T3), or natural desiccated thyroid (NDT) is a rational and increasingly evidence-supported consideration.
A 2025 study found that clinical guidelines from the American Thyroid Association, European Thyroid Association, and British Thyroid Association all remain conservative about routine T3 use, despite the appropriateness of treating with T3 in symptomatic hypothyroid patients with poor quality of life on T4 alone.
Some patients also report significant improvements on natural desiccated thyroid, which contains both T4 and T3 in roughly physiological proportions.
3. Address gut health directly
Given the strong evidence for the gut-thyroid axis, supporting your gut health is not a sideline recommendation — it is a direct intervention in thyroid hormone metabolism.
Specific steps with evidence relevant to the thyroid-gut connection include:
- Probiotic supplementation: A clinical study found that a probiotic combination including Lactobacillus and Bifidobacterium species significantly reduced the levothyroxine dose needed to maintain stable TSH — direct evidence that restoring beneficial gut bacteria improves thyroid hormone bioavailability.
- Addressing intestinal permeability: Reducing dietary triggers of gut inflammation (highly processed foods, refined sugar, potentially gluten in sensitive individuals) and supporting the integrity of your gut barrier with nutrients like zinc, L-glutamine, and short-chain fatty acid-producing foods (fermented vegetables, fiber-rich whole foods) can help improve T4-to-T3 conversion, especially in thyroid patients with autoimmune diseases.
- Selenium: Selenium is required for the activity of deiodinase enzymes. Both the gut microbiome and thyroid hormone conversion depend on adequate selenium status. Multiple clinical trials have shown that selenium supplementation reduces thyroid peroxidase antibodies in Hashimoto’s disease. The gut-thyroid axis research adds another dimension: selenium deficiency impairs the gut’s microbial regulation of thyroid hormone metabolism.
- Taking thyroid medication correctly: Medication absorption is a frequently overlooked variable. Thyroid medications are best absorbed on an empty stomach, 30–60 minutes before food, away from calcium supplements, iron, antacids, and coffee — all of which can impair absorption by 20 to 40%. Ensuring optimal medication absorption is the most basic and immediate step in addressing the conversion-upstream problem.
4. Reduce the inflammatory load
Because systemic inflammation — including the chronic low-grade inflammation driven by Hashimoto’s autoimmunity — directly suppresses deiodinase activity, reducing inflammation is a priority. Anti-inflammatory dietary changes (eating Mediterranean-style, emphasizing omega-3 fatty acids, polyphenol-rich vegetables and fruits, and minimizing ultra-processed foods) are consistently associated with lower levels of inflammation.
The T4-T3 conversion problem is ultimately an argument for more personalized thyroid care— and a clear-eyed acknowledgment that the current standard of care has significant gaps for a meaningful proportion of thyroid patients.
When between 12 and 36 percent of the population carries a genetic variant that impairs the very process that makes standard medication work, that is not a fringe concern. It is a mainstream clinical reality that the field has been slow to incorporate into routine practice. When the gut microbiome — disrupted in the very patient population most affected by thyroid disease — plays a direct role in determining how much T3 thyroid hormone your body produces from the T4 you take, that is not alternative medicine. It is basic physiology.
The research is accumulating. The picture is increasingly clear. The clinical practice is catching up, slowly, unevenly, in pockets of progressive endocrinologists and integrative medicine providers who have been listening to their patients.
If you are on levothyroxine and you do not feel well, you are not alone and you are not imagining it. Push for free T3 testing. Ask about the DIO2 polymorphism. Investigate your gut health. Find a provider like Paloma that is willing to look at the full picture, not just the TSH level.
The medication was never the problem. T4-T3 conversion is!
At Paloma Health, we recognize that treating hypothyroidism requires more than simply normalizing TSH levels. Many patients continue to experience symptoms because standard care overlooks critical factors like T3 levels, thyroid antibody activity, genetic variations, and gut health. That’s why Paloma takes a more comprehensive, personalized approach—looking beyond basic labs to understand how your body is actually functioning.
Our team specializes in identifying and addressing the root causes of persistent hypothyroidism and Hashimoto's thyroiditis symptoms. With advanced thyroid testing, expert providers, and individualized treatment plans—including combination therapy when appropriate—Paloma helps patients optimize their thyroid health, not just manage it. Because feeling “in range” isn’t the same as feeling well—and you deserve both.
Why do I still feel bad if my thyroid labs are normal?
Standard thyroid tests often focus on TSH, which doesn’t measure how much active T3 your body is actually using. You can have normal labs but still have low T3 at the tissue level.
What is T4-to-T3 conversion?
T4 is an inactive thyroid hormone that must be converted into T3, the active form your body uses. This conversion happens in multiple organs and is essential for energy, metabolism, and brain function.
Why doesn’t levothyroxine work for everyone?
Levothyroxine only provides T4, relying on your body to convert it into T3. If that conversion process is impaired, you may not get the full benefit of the medication.
What is the Thr92Ala polymorphism?
It’s a common genetic variation in the DIO2 gene that can reduce your ability to convert T4 into T3. This can lead to persistent symptoms even when lab results look normal.
How common is impaired thyroid hormone conversion?
It’s more common than many people realize, with genetic variants affecting up to one-third of the population. Other factors like stress, inflammation, and nutrient deficiencies can also interfere.
Can gut health affect my thyroid?
Yes, your gut plays a direct role in thyroid hormone metabolism and absorption. An imbalanced microbiome can reduce how much hormone your body activates and reuses.
What is reverse T3 and why does it matter?
Reverse T3 is an inactive form of thyroid hormone that can block T3 from working properly. High levels may contribute to symptoms despite normal standard lab results.
Should I get more than a TSH test?
A complete thyroid panel—including free T3, free T4, and antibodies—provides a much clearer picture of your thyroid health. TSH alone often misses key issues.
What treatments are available if conversion is a problem?
Some patients benefit from adding T3 medication or using combination therapy. Addressing gut health, inflammation, and nutrient deficiencies can also improve conversion.
How can I optimize my thyroid treatment?
Work with a provider who looks at the full picture—not just TSH. Personalized care that considers your symptoms, labs, and underlying factors is key to feeling better.

.webp)
.webp)

%20(1).webp)

%20(1).webp)

%20(1).webp)





