In this article
If you’re over 60 and taking thyroid hormone replacement medication for hypothyroidism, it may be worth asking a surprisingly important question: Can you safely stop taking it, or do you still need it?
A major new study—the RELEASE trial—is putting that question front and center for adults aged 60 and older who take levothyroxine.
For a minority of some older adults, the answer may be yes — especially if the original diagnosis was mild, unclear, or based on asymptomatic borderline or subclinical hypothyroidism. But deprescribing thyroid medicine should always be a careful, clinician-guided decision, because the wrong move can bring back real hypothyroid symptoms and push thyroid levels out of range.
The RELEASE trial was a large Dutch clinical study published in 2026 that investigated whether older adults taking levothyroxine (synthetic thyroid hormone replacement) could safely reduce or stop their medication.
Researchers enrolled adults aged 60 and older who had been on a stable levothyroxine dose for at least 1 year. Participants were gradually tapered off their medication using a structured, step-by-step protocol over the course of about a year.
The study was conducted across 58 general practices in the Netherlands. Patients had regular check-ins with their doctors and repeated TSH and free T4 tests at each stage of dose reduction to monitor thyroid function and safety. The goal was to see who could completely discontinue levothyroxine while maintaining acceptable thyroid hormone levels.
Thyroid treatment often begins with good intentions and then simply continues for years. The RELEASE trial is considered important because, for years, millions of people—especially older adults—have been prescribed levothyroxine, often for mild or borderline thyroid issues. But once started, it’s usually taken for life.
The RELEASE trial challenged that assumption. It’s the largest, most structured study ever to ask: Do some people still need this medication—or could they safely stop it?
This matters because thyroid hormone needs can change with age, and some older adults may be taking more levothyroxine than they now need.
This is why deprescribing is getting more attention: a newer approach encourages doctors to reassess whether levothyroxine is still helpful, particularly in people with mild or subclinical hypothyroidism, unclear diagnoses, or long-term stable use on low doses.
The results of the RELEASE trial are both hopeful and a reality check.
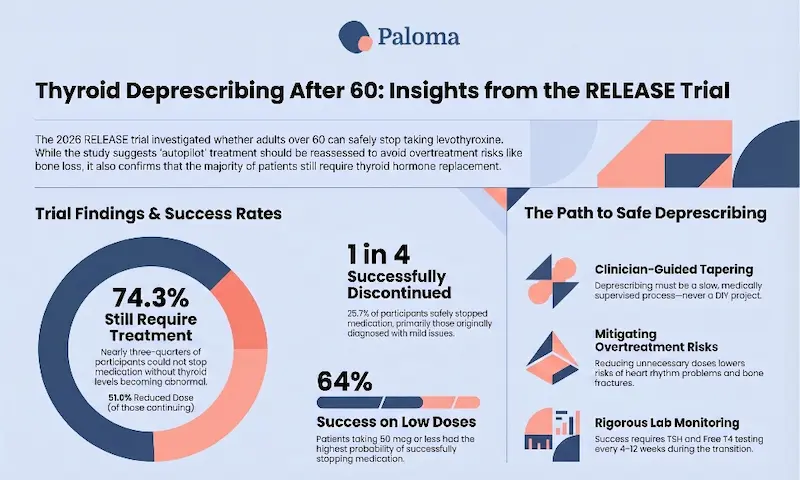
- About 1 in 4 people (25.7%) were deemed a “success” and able to stop levothyroxine after a slow, carefully monitored taper.
- Nearly 3 out of 4 people could NOT stop and needed to stay on the medication.
- People on lower doses (50 mcg or less) had a better chance of stopping—about 64% were able to stop.
- Among those who did not stop, almost 51% had a reduced dose at final follow-up, which can still be beneficial. Almost 8% had an increased dose.
- According to the research, stopping or reducing medication did not worsen quality of life.
Here’s a look at some controversial aspects of the RELEASE trial and its findings.
Definition of “success”
The study considered people “successful” if their TSH remained below 10—even though many doctors—and most thyroid experts—do not consider a TSH of 10 to be evidence of adequate thyroid function. Most laboratories define the upper limit of the normal TSH reference range somewhere between 4.0 and 5.5. Using a threshold of 10 as the criterion for “successful” discontinuation means that a substantial proportion of those classified as having succeeded may in fact be mildly hypothyroid.
That means:
- Some patients identified as “successful” patients may still have mild hypothyroidism
- Using stricter standards, the true success rate may be closer to 12–13%
Reason for treatment
Here’s a big problem: A major limitation of the study is that the researchers didn’t know why many participants were originally put on levothyroxine.
The deprescribing process was done by providers who were essentially “working blind.” Was the patient started on levothyroxine for overt hypothyroidism with elevated TSH and obvious symptoms? Did the patient have autoimmune Hashimoto’s thyroiditis? Did they have subclinical hypothyroidism that has since resolved spontaneously? Did they have a borderline TSH level and nonspecific symptoms? The RELEASE trial hasn’t answered these questions.
The deprescribing process isn’t easily replicated
Deprescribing in the study required:
- Multiple doctor visits
- Lab tests every 6–12 weeks
- Careful dose reductions over months
That’s a lot—especially for older adults managing other health conditions —and is both difficult and costly to replicate in the typical healthcare environment.
Tricky symptoms
Fatigue, weight gain, brain fog? They could be:
- Thyroid-related
- Aging
- Sleep issues
- Heart disease
- Depression
This makes it very easy to misinterpret symptoms—either blaming the thyroid when it’s not the cause, or missing a real issue, or mistakenly attributing thyroid symptoms to other causes.
Deprescribing is not just abruptly stopping a medication. It’s a planned, supervised reduction or discontinuation of a medication that may no longer offer enough benefit to justify its risks. With thyroid medication, that usually means carefully lowering the thyroid hormone replacement drug, then checking symptoms and thyroid labs over time.
Theoretically, the goal is not to “take people off thyroid medicine” as a one-size-fits-all rule. It is to ensure the medication still aligns with the patient’s actual thyroid status and health goals.
There are several reasons experts are paying closer attention to thyroid medication use in older adults. One is that mild TSH elevations may not always require treatment in later life, especially when full hypothyroidism is not present. Another is that studies in older adults with subclinical hypothyroidism have found limited symptom benefit from levothyroxine for many patients.
There is also the concern of overtreatment. Too much thyroid hormone can raise the risk of atrial fibrillation and fractures in older adults, and those risks become more important as people age. In practical terms, that means a medication that once seemed harmless may now be contributing to avoidable harm.
Who may be a candidate for deprescribing?
Deprescribing is most often considered when a person is stable, asymptomatic, on a low dose, or unsure why thyroid treatment was started in the first place. It may also be considered when thyroid tests have been only mildly abnormal, and the medication has been continued out of habit rather than a clear and ongoing need.
By contrast, people with clear overt hypothyroidism, prior thyroid surgery, radioactive iodine treatment, or strongly established lifelong replacement needs are usually not good candidates for stopping therapy.
Deprescribing: Pros for patients
The biggest advantage is avoiding unnecessary treatment and the risks that come with too much thyroid hormone. For some older adults, deprescribing may reduce palpitations, lower the risk of bone loss, and simplify a medication routine that has become crowded over time.
There can also be a quality-of-life benefit if a patient no longer has to manage a drug that may not be doing much for them. In a health system where medications are often continued indefinitely, a thoughtful reassessment can be empowering.
If thyroid medication is no longer needed, there are also cost savings to patients.
Deprescribing: Cons for patients
The main downside of deprescribing is that the majority of adults aged 60 and above who are taking levothyroxine still need it, and lowering or stopping it can allow hypothyroidism to return. That can mean fatigue, constipation, brain fog, weight gain, cold intolerance, dry skin, and worsening cholesterol, among other hypothyroidism symptoms.
There is also uncertainty during the transition. Even if a person feels fine at first, thyroid levels can drift over several weeks, which is why follow-up testing is essential. Deprescribing is not a quick experiment; it is a carefully monitored medical process.

The RELEASE trial findings do not mean you should suddenly stop taking your medication. But it does signal a shift:
- Doctors may start reevaluating long-term thyroid treatment, especially if you’re an older adult
- There’s growing recognition that not everyone needs lifelong therapy
- At the same time, the study confirms that most people still need treatment
It also highlights a major gap: There are still no clear guidelines on how to safely stop or reduce thyroid medication.
Stopping suddenly can cause real problems. Any changes should be gradual and medically supervised.
Ask the right questions
Talk to your provider about:
- Why was I originally prescribed this?
- Do I still need the same dose?
- Am I a candidate for a trial reduction?
Understand your odds
Even in the best-case scenario:
- Most people won’t fully come off medication
- But many can still benefit from a lower dose
Expect a process, NOT a quick fix
If you try deprescribing, your provider should provide a cautious deprescribing plan. This usually means checking thyroid function every 4 to 6 weeks after a dose change so that the medication can reach a new steady state in the body. If symptoms return or TSH rises too far, the medication can be restarted or adjusted.
This stepwise approach is part of what makes deprescribing different from simply “trying to quit.” It is a monitored treatment decision with built-in safety checks.
Pay attention to symptoms—but stay objective
If your thyroid treatment is reduced or stopped, patients and families should watch for symptoms that may suggest the body is not making enough thyroid hormone again. Hypothyroidism symptoms may appear gradually over weeks, not all at once, and include:
- Unusual fatigue or low energy. Feeling tired in a new, persistent way, not explained by sleep or activity changes, can be an early clue.
- Brain fog or slowed thinking. Trouble concentrating, forgetfulness, or feeling mentally sluggish may show up before lab results change dramatically.
- Constipation. Slower bowel movements are a classic symptom of hypothyroidism and can be especially noticeable after stopping medication.
- Feeling colder than usual. If the person starts reaching for sweaters when others are comfortable, that can be a clue.
- Weight gain or fluid retention. Some people notice a slow increase in weight, puffiness, or bloating.
- Dry skin, brittle hair, and breaking nails. Skin may feel rougher, hair may become drier, thinner, or more fragile, and nails may break more easily.
- Muscle aches, weakness, or stiffness. These symptoms can be subtle and are sometimes mistaken for aging or arthritis.
- Low mood or loss of motivation. Depression-like symptoms can overlap with hypothyroidism, making them easy to miss.[7]
- Slower heart rate or reduced exercise tolerance. Some people notice they tire more easily when walking or going up stairs.[4][7]
- Menstrual changes. For people still menstruating, cycles may become heavier or irregular
- Sleep changes. Sleep may feel less restorative.[7]
A few symptoms are more concerning and should be reported quickly: marked swelling, severe weakness, significant confusion, chest symptoms, or a rapid worsening of fatigue or mental slowing. People with heart disease should be especially careful, because thyroid hormone changes can affect heart function and rhythm.
In general, remember that not every symptom is thyroid-related, and decisions should be based on both lab tests AND symptoms.
Think long-term
Even if you successfully stop or reduce your dosage, your thyroid function can decline with age, due to age-related fibrosis, nodular changes, and functional decline. Will a patient who successfully discontinues at age 70 with a TSH of 5.5 be frankly hypothyroid by age 73? Will they know it? Will their provider monitor them? Will symptoms be recognized? The RELEASE trial has no recommendations for addressing these questions, and current guidelines — which do not address deprescribing at all — certainly do not either.
As a result, even if you reduce your dosage or stop taking thyroid medication, you should continue with ongoing thyroid monitoring, as you may need to possibly restart treatment or increase your dosage again later.

The findings from the RELEASE trial arrive at a time when clinical evidence, health policy, and healthcare economics are closely intertwined. That means that some pressure to deprescribe is not just possible, but quite likely. While thyroid hormone medication itself is relatively inexpensive, it is one of the most widely prescribed drugs in the United States. When multiplied across tens of millions of patients—along with routine thyroid lab testing, follow-up visits, and long-term management—the total cost becomes very significant.
For insurers, pharmacy benefit managers (PBMs), and Medicare, research suggesting that a subset of older adults have subclinical hypothyroidism or may not need ongoing treatment represents an opportunity for “medication optimization.” In today’s cost-conscious healthcare environment, that kind of data is unlikely to go unnoticed.
The incentive for deprescribing thyroid hormone in older adults is unlikely to come in the form of a blunt directive to stop treatment categorically. Instead, it will more likely take the form of subtle but powerful administrative pressures. These can include increased prior authorization requirements for refills, prompts during Medicare Annual Wellness Visits to reassess necessity, and quality metrics tied to reducing “potentially unnecessary medications.” Within Medicare Advantage and Accountable Care Organizations, financial incentives tied to performance metrics can further encourage providers to reevaluate ongoing prescriptions. On paper, these measures are presented as patient safety and quality-improvement efforts. In practice, they can shift the burden onto clinicians and patients to justify continued therapy, even when it remains necessary and appropriate for the patients.
The challenge is that the careful, highly monitored deprescribing process used in the RELEASE trial is unlikely to be followed in real-world practice by most providers. The study involved gradual dose reductions, frequent thyroid-stimulating hormone (TSH) and other lab testing, and shared decision-making—conditions that are difficult to replicate in routine care. Without that level of oversight, deprescribing can become clinically risky, particularly for older adults with underlying heart disease, cognitive concerns, or long-standing hypothyroidism. Just as overtreatment carries risks, so does undertreatment or abrupt withdrawal.
Another key issue is that many patients—and even their current providers—may not know why thyroid hormone replacement therapy was originally prescribed. Hypothyroidism is not a single condition; it can stem from permanent thyroid failure, transient dysfunction, medication exposure, autoimmune disease, or even a one-time borderline lab result. Population-level efforts to reduce prescribing do not account for these nuances, creating a real risk that appropriate, necessary treatment could be discontinued based on incomplete information.
For patients, the takeaway is not to be alarmed—but to be prepared.
If the topic of deprescribing comes up, it should trigger a thoughtful, individualized discussion—not a quick decision driven by policy or cost concerns. You can advocate for yourself by asking key questions:
- What was the original diagnosis?
- Is my thyroid condition permanent?
- What happens if we reduce my dose?
- How will we monitor the process?
A safe deprescribing plan, if appropriate, should always include gradual adjustments, regular lab testing, and close attention to symptoms.
At the same time, clinicians and professional organizations should play a critical role in shaping how this evolves. Ideally, groups like the American Thyroid Association and the Endocrine Society will advocate for clear, evidence-based guidelines to ensure that any deprescribing efforts remain patient-centered and clinically sound. Their involvement can help prevent cost-driven policies from overriding individualized care.
Ultimately, the broader deprescribing movement—supported by agencies such as the Centers for Medicare & Medicaid Services and the Agency for Healthcare Research and Quality—has the potential to improve care by reducing unnecessary medications. But with thyroid hormone replacement, the margin for error is quite narrow. The goal should not simply be fewer prescriptions—it should be the right treatment, for the right patient, at the right time.
If you’re over 60, the real question is not whether hypothyroidism treatment should always be deprescribed — it’s whether your treatment is still appropriate for your current health status, at any age.
The RELEASE trial is a wake-up call: some people may be taking thyroid medication they no longer need. For some people, especially those with mild or uncertain hypothyroidism, the answer may be to lower or stop medication – under medical supervision.
The reality is, however, that most people still do have overt hypothyroidism, need treatment, and medication remains the right choice.
So, the real takeaway isn’t “stop your medication.” It’s that your thyroid treatment shouldn’t be on autopilot. Regular re-evaluation, personalized care, and informed decision-making are now more important than ever.
The key is to reassess, not assume. That’s the safest way to balance the possible benefits of deprescribing against the very real risk of thyroid symptoms returning.[3][1]
Paloma Health is uniquely positioned to help patients safely evaluate whether thyroid hormone therapy should be continued or carefully reduced. With affordable, convenient at-home thyroid testing and access to clinicians who specialize in thyroid care, Paloma makes it easier to monitor key thyroid lab values throughout any deprescribing process—without the barriers of frequent in-office visits or high costs.
Just as importantly, Paloma’s model supports ongoing, personalized care, allowing for gradual dose adjustments, symptom tracking, and shared decision-making at every step. This kind of structured, closely supervised approach mirrors the rigor of clinical trials and helps ensure that any attempt to deprescribe is done safely, thoughtfully, and based on each patient’s individual history—not on broad population-level assumptions.

Can some people really stop taking thyroid medication after 60?
Yes, but only a minority—about 25% in the study—were able to stop safely, usually under careful medical supervision.
Should I stop my thyroid medication on my own?
No. Stopping suddenly can cause symptoms to return and may lead to serious health issues—always work with your healthcare provider.
Who is most likely to stop medication successfully?
People on low doses, with mild or unclear diagnoses, or those who were treated for borderline thyroid issues, may have a better chance.
What happens if I stop but still need the medication?
Symptoms such as fatigue, weight gain, brain fog, and cold intolerance can recur, and your thyroid levels may become abnormal.
Can I at least lower my dose instead of stopping completely?
Yes, many people benefit from dose reduction even if they can’t fully stop.
Why are doctors reconsidering thyroid treatment in older adults?
Because mild thyroid abnormalities may not always need treatment later in life, and too much medication can increase risks like fractures or heart issues.
What is “deprescribing”?
It’s a careful, step-by-step process of reducing or stopping a medication that may no longer be necessary, done under medical supervision.
How is thyroid medication safely reduced?
Gradually, over months, with lab testing every 4–6 weeks and monitoring for symptoms.
What symptoms should I watch for if my dose is reduced?
Fatigue, brain fog, constipation, feeling cold, weight gain, and dry skin are common signs your body may need more thyroid hormone.
What’s the biggest takeaway from this research?
Don’t assume your treatment should stay the same forever—regularly reassess with your doctor to make sure it’s still right for you.

.webp)
.webp)
%20copy.webp)

%20(1).webp)








